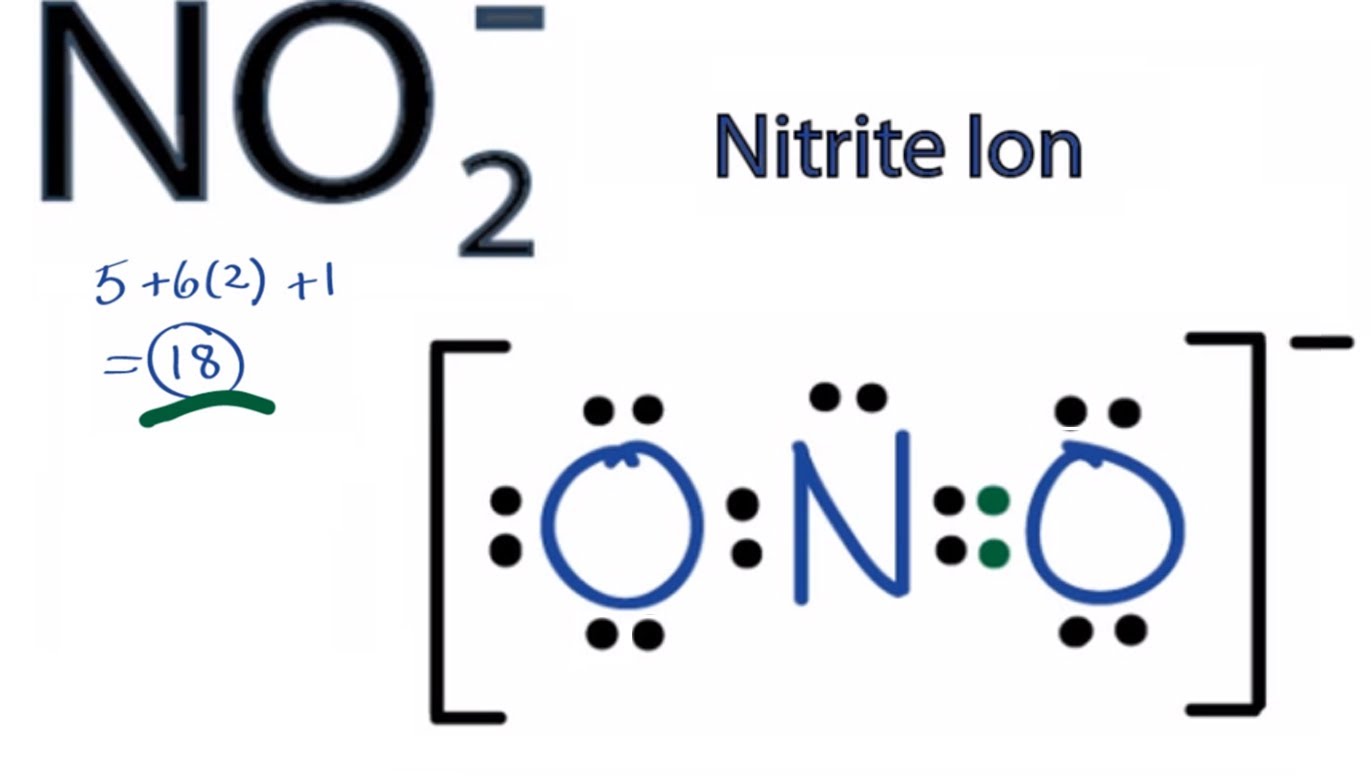
The convention for ions is to enclose the structure in parentheses and indicate the net charge in the upper right corner. If all atoms in the second period and above have at least one octet, and no atom in the second period exceeds an octet, and the total number of electrons in bonds and lone pairs equals the total number of available valence electrons, then we have produced a valid Lewis structure. 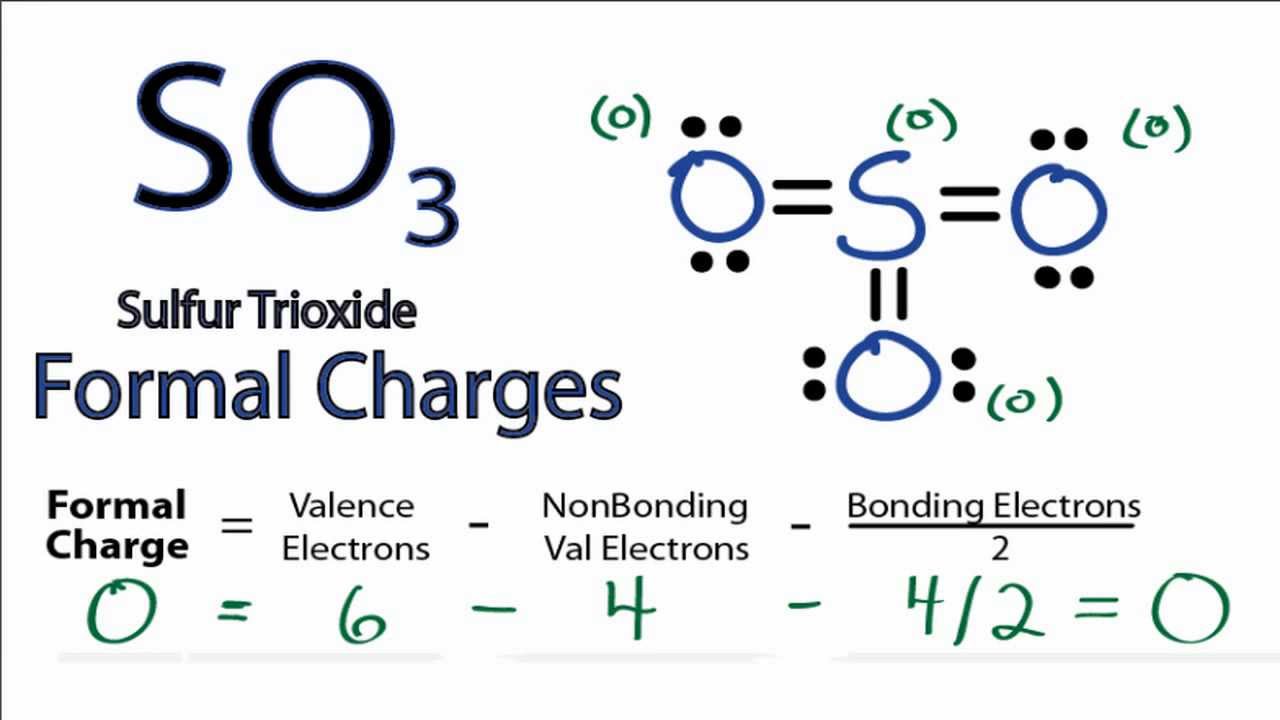
Carbon and nitrogen are second-period elements that commonly form double and triple bonds as central atoms, and oxygen as the peripheral atom is often in a double bond with the central atom. If no more electrons are available and the central one does not yet have a full octet, a lone pair on a peripheral atom can be pushed into a second (or third) bond with the central atom. How to calculate the formal charges on NO3 atoms The formal charges can be calculated using the formula given below: The formal charge of an atom valence electrons of an atom non-bonding electrons (bonding electrons) The valence electrons (V.E) of an atom are the total number of electrons present in its valence shell. Never exceed an octet for an atom of period 2! For periods 3 and larger, the atoms are large enough to accommodate more than one octet in their valence bond shell. Add the remaining pairs of electrons to the central atom so your octet is complete (if it isn’t already).Note that this does not apply to hydrogen H, which can only accommodate a duo (2 e – ). Add electrons as lone nonbonding pairs around peripheral atoms so they have octets (eight electrons total).Inevitably, there will be cases where it is possible to draw more than one skeletal structure.ĭraw bonds as lines between atoms. Another good way to choose is to choose the least electronegative atom. Given a formula, the central atom is typically the first atom (eg ClF 4 ), although this convention is not always followed (eg HNO 3 ). Hydrogen (H) and fluorine (F) each have a valence of 1, and generally these will not be central atoms (bonded to more than one atom). 
Choose a central atom (we will take as examples small molecules for which there is only one central atom, and the other atoms, the peripheral atoms, are all attached to the central atom). Which will help us decide how the atoms will bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
